
An easy calculation gave the charge-to-mass ratio. That value was then inserted along with the other values into the deflection formulas shown above. Since Thomson knew both the electrical and magnetic field strengths as well as the amount of deflection, he could easily solve for the velocity. Then, the mass, charge and both lengths cancel, leaving us with: (Several years previous to 1897, Thomson had measured the cathode rays' velocity, but he grew to distrust the results.) However, both equations can be used as a ratio if the deflections by the two fields are made to be equal. Notice that both equations depend on knowing the velocity of the cathode ray. A different experiment would have to be carried out. However, the two formulas above could not give either the charge or the mass by itself. 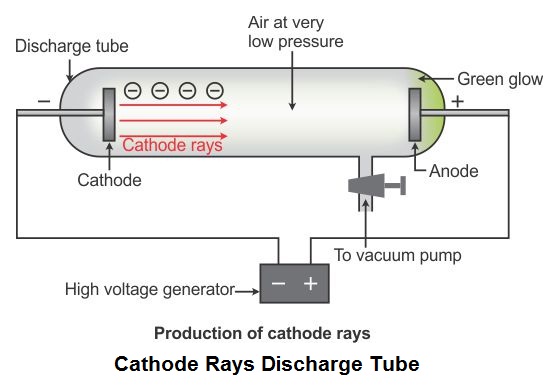
Just below are GIFs of each formula.īy carrying out the experiments and measuring the proper values, he could calculate what the charge-to-mass (e/m) ratio was for the cathode ray. Thomson had developed formulas based on the deflection of the cathode ray by the electric field and by the magnetic field. (May 1996: This will be made a link when that section is written.)įor a fuller discussion of the below, please see "The Discovery of Subatomic Particles" by Steven Weinberg. Knowledge of the value of 'e' or of 'm' would be needed to get to the other once you knew e/m, which Thomson did know.Įlsewhere you will find discussion of how the value for 'e,' the charge on the electron was determined. The e/m ratio is important because that is as far as Thomson could get with his cathode ray tubes. Reversing the above figures and using grams rather than kilograms gives a value of 5.686 x 10 -9 g/C. Many textbooks and articles use the m/e ratio, that is the mass-to-charge ratio. It is about one meter in length and was made entirely by hand. The image below of a CRT used by Thomson in his experiments. Only the end of the CRT can be seen to the right-hand side of the picture. The electrons emitted by a heated cathode move from atom to another atom in the form of an electric. Thomson and a cathode ray tube from around 1897, the year he announced the discovery of the electron. The cathode ray tube serves the function of conversion of electrical signal into visual display. Often, books round off the 1.759 portion to 1.76. Cathode ray tube experiment is the result of Sir J.J Thomson discovery of electron, full name of J.J Thomson is Joseph John Thomson. Usually, grams are used rather than kilograms giving a numerical value of 1.759 x 10 8. Therefore, the modern value for the e/m ratio is 1.759 x 10 11 C/kg. 
The modern value for the charge on the electron (to four significant places) is 1.602 x 10 -19 coulombs and the electrons mass is 9.109 x 10 -31 kilograms. The cathode rays bend toward the positive pole, confirming that cathode rays is negatively charged.Į/m ratio stands for charge-to-mass ratio of the electron. Thomson is the first individual to succeed in deflecting the cathode ray with an electrical field. The discovery of this effect in 1855 predates by some ten years the unification of electricity and magnetism by James Clerk Maxwell.Ĥ) Although there was some speculation that the cathode rays were negatively charged, it is not shown to be true by experiment until 1895, just two years before Thomson announces the electron.ĥ) J.J. cience lecturers who traveled from town to town in the mid nineteenth century delighted audiences by showing them the ancestor of the neon sign. This showed that the cathode ray carried energy and could do work.ģ) The cathode ray is deflected from a straight line path by a magnetic field, suggesting that the two were related in some way. Thomson was able to deflect the cathode ray towards a positively charged plate deduce that the particles in the beam were negatively charged. 'There is no other branch of physics which affords us so promising an opportunity of penetrating the secret of electricity.' - J.J. This showed that the cathode rays traveled in straight lines.Ģ) The cathode ray can push a small paddle wheel up an incline, against the force of gravity. Thomson was an English physicist who began experimenting with cathode ray tubes. What should my next move be? The project ran properly before the Netbeans upgrade, but not after, and no code was changed.There were a number of results gathered over the years by cathode ray tube researchers.ġ) If an object is placed in the path of the cathode ray, a shadow of the object is cast on the glowing tube wall at the end. Implementing the cathode ray experiment was undertaken by J. I’m guessing this has something to do with flatpak being sandboxed, but I’m still not sure if even that is the issue. 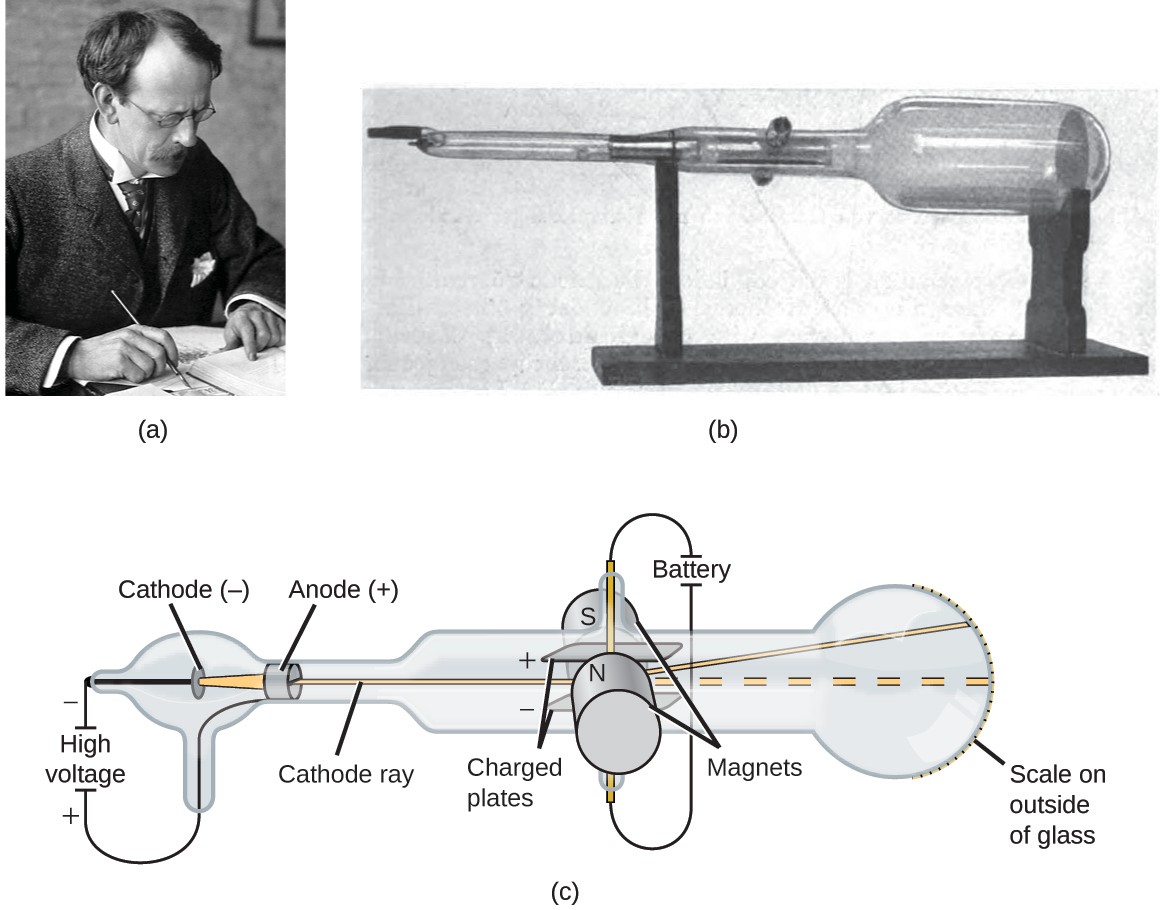
I changed the Java platform from JDK19, to JDK20, however, the Netbeans info center seems to report java 11, despite my java -version being: java version "20". This caused an error in one of the projects I’m working on, and for some reason, in the media player (I suspect some backend issue, not a code issue).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
